Abstract
There are different opinions around the World regarding the zoonotic capability of H3N8 equine influenza viruses. In this report, we have tried to summarize the findings of different research and review articles from Chinese, English, and Mongolian Scientific Literature reporting the evidence for equine influenza virus infections in human beings. Different search engines i.e. CNKI, PubMed, ProQuest, Chongqing Database, Mongol Med, and Web of Knowledge yielded 926 articles, of which 32 articles met the inclusion criteria for this review. Analyzing the epidemiological and Phylogenetic data from these articles, we found a considerable experimental and observational evidence of H3N8 equine influenza viruses infecting human being in different parts of the World in the past. Recently published articles from Pakistan and China have highlighted the emerging threat and capability of equine influenza viruses for an epidemic in human beings in future. In this review article we have summarized the salient scientific reports published on the epidemiology of equine influenza viruses and their zoonotic aspect. Additionally, several recent developments in the start of 21st century, including the transmission and establishment of equine influenza viruses in different animal species i.e. camels and dogs, and presumed encephalopathy associated to influenza viruses in horses, have documented the unpredictable nature of equine influenza viruses. In sum up, several reports has highlighted the unpredictable nature of H3N8 EIVs highlighting the need of continuous surveillance for H3N8 in equines and humans in contact with them for novel and threatening mutations.
Keywords:
zoonotic; equine influenza; viruses; epidemic; epidemiology
Resumo
Existem diferentes opiniões em todo o mundo a respeito da capacidade zoonótica dos vírus da influenza equina H3N8. Neste relatório, tentamos resumir os resultados de diferentes pesquisas e artigos de revisão da literatura científica chinesa, inglesa e mongol relatando as evidências de infecções pelo vírus da influenza equina em seres humanos. Diferentes mecanismos de busca, como CNKI, PubMed, ProQuest, Chongqing Database, Mongol Med e Web of Knowledge geraram 926 artigos, dos quais 32 atenderam aos critérios de inclusão para esta revisão. Analisando os dados epidemiológicos e filogenéticos desses artigos, encontramos uma considerável evidência experimental e observacional de vírus da influenza equina H3N8 infectando seres humanos em diferentes partes do mundo no passado. Artigos publicados recentemente no Paquistão e na China destacaram a ameaça emergente e a capacidade dos vírus da influenza equina para uma epidemia em seres humanos no futuro. Neste artigo de revisão, resumimos os relatórios científicos relevantes publicados sobre a epidemiologia dos vírus da influenza equina e seu aspecto zoonótico. Além disso, vários desenvolvimentos recentes no início do século 21, incluindo a transmissão e estabelecimento de vírus da influenza equina em diferentes espécies animais, ou seja, camelos e cães, e presumida encefalopatia associada aos vírus da influenza em cavalos, documentaram a natureza imprevisível dos vírus da influenza equina. Em suma, vários relatórios destacaram a natureza imprevisível de H3N8 EIVs destacando a necessidade de vigilância contínua para H3N8 em equinos e humanos em contato com eles para novas mutações ameaçadoras.
Palavras-chave:
zoonótico; influenza equina; vírus; epidemia; epidemiologia
1. Introduction
Equine influenza is a common respiratory infection of horses caused by an orthomyxovirus of the genus influenza A. Influenza A viruses can be divided into sub-types based on antigenic reactivity of the surface glycoproteins, the haemagglutinin (H1– H16) and neuraminidase (N1–N9) molecules (Fouchier et al., 2005FOUCHIER, R.A., MUNSTER, V., WALLENSTEN, A., BESTEBROER, T.M., HERFST, S., SMITH, D., RIMMELZWAAN, G.F., OLSEN, B. and OSTERHAUS, A.D., 2005. Characterization of a novel influenza A virus hemagglutinin subtype (H16) obtained from black-headed gulls. Journal of Virology, vol. 79, no. 5, pp. 2814-2822. http://dx.doi.org/10.1128/JVI.79.5.2814-2822.2005. PMid:15709000.
http://dx.doi.org/10.1128/JVI.79.5.2814-...
). The viruses currently circulating in horses are of the H3N8 sub-type. Although influenza viruses were first isolated in the 1930s, we still have much to learn about this pathogen. Influenza A viruses have only eight RNA gene segments encoding between them 11 or 12 proteins, yet the existence of two of these proteins were only reported in this decade (Chen et al., 2001CHEN, W., CALVO, P.A., MALIDE, D., GIBBS, J., SCHUBERT, U., BACIK, I., BASTA, S., O’NEILL, R., SCHICKLI, J., PALESE, P., HENKLEIN, P., BENNINK, J.R. and YEWDELL, J.W., 2001. A novel influenza A virus mitochondrial protein that induces cell death. Nature Medicine, vol. 7, no. 12, pp. 1306-1312. http://dx.doi.org/10.1038/nm1201-1306. PMid:11726970.
http://dx.doi.org/10.1038/nm1201-1306...
, Wise et al., 2009WISE, H.M., FOEGLEIN, A., SUN, J., DALTON, R.M., PATEL, S., HOWARD, W., ANDERSON, E.C., BARCLAY, W.S. and DIGARD, P., 2009. A complicated message: identification of a novel PB1-related protein translated from influenza A virus segment 2 mRNA. Journal of Virology, vol. 83, no. 16, pp. 8021-8031. http://dx.doi.org/10.1128/JVI.00826-09. PMid:19494001.
http://dx.doi.org/10.1128/JVI.00826-09...
). Influenza is a classic example of a (re-)emerging infection. Vaccines against influenza have been used in man since the 1940s (Francis Junior et al., 1945FRANCIS JUNIOR, T., SALK, J.E., PEARSON, H.E. and BROWN, P.N., 1945. Protective effect of vaccination against induced influenza A. The Journal of Clinical Investigation, vol. 24, no. 4, pp. 536-546. http://dx.doi.org/10.1172/JCI101633. PMid:16695243.
http://dx.doi.org/10.1172/JCI101633...
), and became available for use in horses 20 years later. However, the existence of a reservoir of virus in aquatic birds and the highly variable nature of the virus means that influenza defies worldwide eradication. This review focuses on some of the obstacles, highlighted by recent events that must still be surmounted if improvements are to be made in the control of equine influenza, while some areas for future research are suggested.
Influenza A virus-like-illnesses have been recognized in horses since at least 1299, with speculation that earlier outbreaks of equid diseases could have also been due to influenza A viruses (Gibbs and Anderson, 2010GIBBS, E.P.J. and ANDERSON, T.C., 2010. Equine and canine influenza: a review of current events. Animal Health Research Reviews, vol. 11, no. 1, pp. 43-51. http://dx.doi.org/10.1017/S1466252310000046. PMid:20426896.
http://dx.doi.org/10.1017/S1466252310000...
, Morens and Taubenberger, 2010b). A particularly well-documented influenza-like epizootic occurred among USA horses during 1872, causing widespread damage to transportation and commerce (Morens and Taubenberger, 2010aMORENS, D.M. and TAUBENBERGER, J.K., 2010a. An avian outbreak associated with panzootic equine influenza in 1872: an early example of highly pathogenic avian influenza? Influenza and Other Respiratory Viruses, vol. 4, no. 6, pp. 373-377. http://dx.doi.org/10.1111/j.1750-2659.2010.00181.x. PMid:20958931.
http://dx.doi.org/10.1111/j.1750-2659.20...
). Morens and Taubenberger (2010a)MORENS, D.M. and TAUBENBERGER, J.K., 2010a. An avian outbreak associated with panzootic equine influenza in 1872: an early example of highly pathogenic avian influenza? Influenza and Other Respiratory Viruses, vol. 4, no. 6, pp. 373-377. http://dx.doi.org/10.1111/j.1750-2659.2010.00181.x. PMid:20958931.
http://dx.doi.org/10.1111/j.1750-2659.20...
have observed that this 1872 outbreak could have been evidence of avian influenza virus cross-species infections but as the first influenza A viruses were not discovered until the 1930s, the etiology of the 1872 epizootic is unknown. Since the 1930s, only two major subtypes of equine influenza viruses (EIV) have been detected in ill horses: H7N7 (first called A/equi-1) (Nemoto et al., 2012NEMOTO, M., YAMANAKA, T., BANNAI, H., TSUJIMURA, K., KONDO, T. and MATSUMURA, T., 2012. Development of a reverse transcription loop-mediated isothermal amplification assay for H7N7 equine influenza virus. The Journal of Veterinary Medical Science, vol. 74, no. 7, pp. 929-931. http://dx.doi.org/10.1292/jvms.11-0541. PMid:22362254.
http://dx.doi.org/10.1292/jvms.11-0541...
) and H3N8 (first called A/equi-2) (Van Maanen and Cullinane, 2002VAN MAANEN, C. and CULLINANE, A., 2002. Equine influenza virus infections: an update. The Veterinary Quarterly, vol. 24, no. 2, pp. 79-94. http://dx.doi.org/10.1080/01652176.2002.9695127. PMid:12095083.
http://dx.doi.org/10.1080/01652176.2002....
). The last H7N7 viruses were isolated in the late 1970s (Van Maanen and Cullinane, 2002VAN MAANEN, C. and CULLINANE, A., 2002. Equine influenza virus infections: an update. The Veterinary Quarterly, vol. 24, no. 2, pp. 79-94. http://dx.doi.org/10.1080/01652176.2002.9695127. PMid:12095083.
http://dx.doi.org/10.1080/01652176.2002....
, Krueger et al., 2014KRUEGER, W.S., HEIL, G.L., YOON, K.J. and GRAY, G.C., 2014. No evidence for zoonotic transmission of H 3 N 8 canine influenza virus among US adults occupationally exposed to dogs. Influenza and Other Respiratory Viruses, vol. 8, no. 1, pp. 99-106. http://dx.doi.org/10.1111/irv.12208. PMid:24237615.
http://dx.doi.org/10.1111/irv.12208...
), with only variants of H3N8 viruses causing sporadic outbreaks since then. Except for recent H3N8 EIV variants which have caused outbreaks in dogs (Crawford et al., 2005CRAWFORD, P.C., DUBOVI, E.J., CASTLEMAN, W.L., STEPHENSON, I., GIBBS, E., CHEN, L., SMITH, C., HILL, R.C., FERRO, P., POMPEY, J., BRIGHT, R.A., MEDINA, M.J., JOHNSON, C.M., OLSEN, C.W., COX, N.J., KLIMOV, A.I., KATZ, J.M. and DONIS, R.O., 2005. Transmission of equine influenza virus to dogs. Science, vol. 310, no. 5747, pp. 482-485. http://dx.doi.org/10.1126/science.1117950. PMid:16186182.
http://dx.doi.org/10.1126/science.111795...
), and possibly our case report of an H3N8 infection in a camel (Yondon et al., 2014YONDON, M., ZAYAT, B., NELSON, M.I., HEIL, G.L., ANDERSON, B.D., LIN, X., HALPIN, R.A., MCKENZIE, P.P., WHITE, S.K., WENTWORTH, D.E. and GRAY, G.C., 2014. Equine influenza A (H3N8) virus isolated from Bactrian camel, Mongolia. Emerging Infectious Diseases, vol. 20, no. 12, pp. 2144-2147. http://dx.doi.org/10.3201/eid2012.140435. PMid:25418532.
http://dx.doi.org/10.3201/eid2012.140435...
), recent EIV epizootics have not been associated with spill-over to non-equid species. In this report, we sought to review the English, Chinese, and Mongolian scientific literature for evidence that EIV infections have occurred in man. The Chinese and Mongolian literature were thought to be very relevant because in China’s autonomous region of Inner Mongolia and in the country of Mongolia, large numbers of horses have close contact with man. Considering the recent outbreaks and mutation of equine influenza viruses (Khan et al., 2018KHAN, A., MUSHTAQ, M.H., AHMAD, M.U.D., NAZIR, J., FATIMA, Z., KHAN, A. and FAROOQI, S.H., 2018. Molecular epidemiology of the two internal genes of equine influenza H3N8 virus isolated in Pakistan 2015-16. Pakistan Veterinary Journal, vol. 38, no. 1, pp. 96-100.) in developing countries, the current systematic review article summarizes the findings of published literature to investigate and predict the epidemiological zoonotic aspect of equine influenza viruses in future.
2. Materials and Methods
2.1. Equine population globally
Globally decline in the equine population have been reported at a rate of one million per year since 2009, such that 60 million horses were listed in 2009 when this number dropped to 58.5 million in 2011. While the donkeys and mules population have increased, a total of 54 million donkeys and mules have listed, a number that is inching up towards the horse’s population. The United States of America is leading the World’s Horses Population having 10 million horses followed by China, Mexico, Brazil and Argentina, each having more than 3.5 million horses (FAO Global Horse population report 2006).
2.2. Searching articles
An inclusive literature search was conducted in CNKI, Chongqing Database, Mongol Med, ProQuest, PubMed, Web of Knowledge and Wanfang Data using the following key search terms, respectively: “[Country Name] and [equine influenza] and [prevalence] and [outbreak or epidemic or equine influenza zoonosis]. Additionally several studies were identified manually of references noted in publications. Non-indexed national reports and communication with experts available on internet were also included (Horton, 2013HORTON, R., 2013. Metrics for what? The Lancet, vol. 381, pp. S1-S2. http://dx.doi.org/10.1016/S0140-6736(13)61256-8.
http://dx.doi.org/10.1016/S0140-6736(13)...
, Arthur, 2014ARTHUR, M., 2014. Institute for health metrics and evaluation. Nursing Standard, vol. 28, no. 42, pp. 32. http://dx.doi.org/10.7748/ns.28.42.32.s33. PMid:25005408.
http://dx.doi.org/10.7748/ns.28.42.32.s3...
). Article abstracts and titles were reviewed for the relevance and the following reliable data were extracted from full length articles or abstracts: equine influenza prevalence, epidemic frequencies, studied population and analysis type (meta-analysis, review article, surveillance studies, outbreak investigations and other). Research studies conducted in non-representative population, with less than 100 sample size and articles published prior to 2000 were not included in this systematic review for analysis.
2.3. Quality score
For selection of studies a multi-objective decision-analysis approach (Jong and Stone, 2006JONG, N.K. and STONE, P., 2006. Kernel-Based Models for Reinforcement Learning. In: Proceedings of the ICML-06 Workshop on Kernel Methods in Reinforcement Learning, 25-29 January 2006, Pittsburgh, Pennsylvania, USA. New Jersey: John Wiley & Sons., Hammond et al., 2002HAMMOND, J., KEENEY, R. and RAIFFA, R., 2002. A practical guide to making better life decisions. New York: Broadway Books.) was used to derive 0-10 score for each article, based on three basic measure: sample size, year of analysis, and how well the study data could be extrapolated to the general population. The sample size log was scaled as 0-10: where studies were given score 10 out of 10 if the sample size is greater than 1000, studies conducted from 2000 to 2010 received score of 6, 2011-2015 score of 8 and >2016 a score of 10. The final score was estimated using a weighting of 50% for the extrapolation score and 30% for sample size and year of study. Studies with highest scores were selected for base assumption. Studies conducted based on single clinics were excluded from the base estimates due to representation of non-representative prevalence of the area. In case of insufficient data available to estimate the equine influenza range in a country, then data from neighboring countries or countries in the region were included.
2.4. Analysis of data
Statistical data was analyzed using SPSS version 22.00 and sequence data was analyzed using the bioinformatics software known as Geneious version 11.0 for Phylogenetic tree construction and amino acid alignment.
3. Results
3.1. Epidemiology of Equine Influenza
3.1.1. Incubation period
Equine Influenza (EI) is characterized by incubation period of 5 days and infective period of 14 days. In field during server outbreaks an incubation period of 3 days have been reported in susceptible equine population. In naïve equines this period could be less than a day (Cullinane et al., 2010CULLINANE, A., ELTON, D. and MUMFORD, J., 2010. Equine influenza–surveillance and control. Influenza and Other Respiratory Viruses, vol. 4, no. 6, pp. 339-344. http://dx.doi.org/10.1111/j.1750-2659.2010.00176.x. PMid:20958927.
http://dx.doi.org/10.1111/j.1750-2659.20...
) and virus excretion might persist up to 10 days (Kirkwood, 1998KIRKWOOD, C., 1998. Strategic decision making multiobjective decision analysis with spreadsheets. The Journal of the Operational Research Society, vol. 49, no. 1, pp. 96-97. http://dx.doi.org/10.1057/palgrave.jors.2600003.
http://dx.doi.org/10.1057/palgrave.jors....
). Shedding of the virus mostly occurs in earlier stages of infection when most pronounced coughing is observed. In vaccinated or immune equines no clinical signs or rarely mild clinical signs are observed but virus shedding occurs.
3.1.2. Pathogenicity of equine influenza viruses
There are different types of influenza virus (A, B and C) with almost similar genetic makeup (Figure 1), where only influenza type A viruses have higher propensity of crossing species barrier. The other two type B and C are mostly found in Humans. Influenza-A viruses have been isolated from different species including birds, horses, humans, swine, dogs and marine mammals. Only a few number of the influenza type A subtypes and their combinations have established in different mammalian species (H7N7 and H3N8 in equines; H1N1, H5N1, H2N2 and H3N2 in humans) (Studdert, 1996STUDDERT, M.J., 1996. Virus infections of equines. Amsterdam: Elsevier., Daly et al., 2011DALY, J.M., MACRAE, S., NEWTON, J.R., WATTRANG, E. and ELTON, D.M., 2011. Equine influenza: a review of an unpredictable virus. Veterinary Journal, vol. 189, no. 1, pp. 7-14. http://dx.doi.org/10.1016/j.tvjl.2010.06.026. PMid:20685140.
http://dx.doi.org/10.1016/j.tvjl.2010.06...
). Influenza A viruses isolated from aquatic birds are thought to be the ancestors of all the influenza sub types existing in rest of the animals such as dogs, humans, horses and pigs. Initially horses were suggested to be the dead end hosts for H3N8 viruses lacking the exchange of the viral genes from other species influenza viruses (Gorman et al., 1991GORMAN, O.T., BEAN, W., KAWAOKA, Y., DONATELLI, I., GUO, Y. and WEBSTER, R., 1991. Evolution of influenza A virus nucleoprotein genes: implications for the origins of H1N1 human and classical swine viruses. Journal of Virology, vol. 65, no. 7, pp. 3704-3714. http://dx.doi.org/10.1128/jvi.65.7.3704-3714.1991. PMid:2041090.
http://dx.doi.org/10.1128/jvi.65.7.3704-...
). However in 2004 an outbreak in racing Greyhounds in United States of America was found to be due to an equine influenza virus (Crawford et al., 2005CRAWFORD, P.C., DUBOVI, E.J., CASTLEMAN, W.L., STEPHENSON, I., GIBBS, E., CHEN, L., SMITH, C., HILL, R.C., FERRO, P., POMPEY, J., BRIGHT, R.A., MEDINA, M.J., JOHNSON, C.M., OLSEN, C.W., COX, N.J., KLIMOV, A.I., KATZ, J.M. and DONIS, R.O., 2005. Transmission of equine influenza virus to dogs. Science, vol. 310, no. 5747, pp. 482-485. http://dx.doi.org/10.1126/science.1117950. PMid:16186182.
http://dx.doi.org/10.1126/science.111795...
). Genetic analysis estimated that the isolates from canines were closely similar to the contemporary viral strains of the equine influenza H3N8 viruses (Crawford et al., 2005CRAWFORD, P.C., DUBOVI, E.J., CASTLEMAN, W.L., STEPHENSON, I., GIBBS, E., CHEN, L., SMITH, C., HILL, R.C., FERRO, P., POMPEY, J., BRIGHT, R.A., MEDINA, M.J., JOHNSON, C.M., OLSEN, C.W., COX, N.J., KLIMOV, A.I., KATZ, J.M. and DONIS, R.O., 2005. Transmission of equine influenza virus to dogs. Science, vol. 310, no. 5747, pp. 482-485. http://dx.doi.org/10.1126/science.1117950. PMid:16186182.
http://dx.doi.org/10.1126/science.111795...
). Furthermore, the equine influenza virus transmission into dogs during a recent outbreak in Australia was reported (Kirkland et al., 2010KIRKLAND, P.D., FINLAISON, D.S., CRISPE, E. and HURT, A.C., 2010. Influenza virus transmission from horses to dogs, Australia. Emerging Infectious Diseases, vol. 16, no. 4, pp. 699-702. http://dx.doi.org/10.3201/eid1604.091489. PMid:20350392.
http://dx.doi.org/10.3201/eid1604.091489...
). There is no clear evidence that canine influenza viruses have caused widespread problems in United Kingdom or other European countries.
3.1.3. Survival and persistence of EIV
The EIVs have a lipid envelope but it could not survive for too long outside the host. Its highly susceptible to ultraviolet light rays if exposed for more than 30 minutes, or if heated at 50°C for 30 min. it could also be deactivated by ether or acid (pH=3) and it is also fragile to high humidity if environmental conditions stays humid for long period (Yadav et al., 1993YADAV, M., UPPAL, P. and MUMFORD, J., 1993. Physico-chemical and biological characterization of A/Equl-2 virus isolated from 1987 equine influenza epidemic in India. International Journal of Animal Sciences, vol. 8, pp. 93-98.). However the EIV can survive on skin, contaminated equipment surface and fabrics for some time. It is reported that virus does not persist mostly in recovered equines but it is also thought that the virus could persist in endemic equine population by low grade and circulates causing occasional small epidemics (Glass et al., 2002GLASS, K., WOOD, J., MUMFORD, J., JESSET, D. and GRENFELL, B., 2002. Modelling equine influenza 1: a stochastic model of within-yard epidemics. Epidemiology and Infection, vol. 128, no. 3, pp. 491-502. http://dx.doi.org/10.1017/S0950268802006829. PMid:12113495.
http://dx.doi.org/10.1017/S0950268802006...
). Information is lacking to report the persistence of EIV in horse carcasses but it could be expected if animal dies during viraemic phase of the disease.
3.1.4. Interspecies transmission of equine influenza viruses
Interspecies transmission of the Influenza A viruses rely on several factors. For these viruses to enter into the host cells, the hemaglutinin (HA) protein needs to bind with sialic acid (SA) receptors on the host cell surface (Gottschalk et al., 1972GOTTSCHALK, A., BELYAVIN, G. and BIDDLE, F., 1972. Glycoproteins as influenza virus haemagglutinin inhibitors and as cellular virus receptors. In: A. Gottschalk, ed. Glycoproteins. Amsterdam: Elsevier.). Viruses being originated from wild aquatic birds binds strongly to the SA site in a 2, 3 linkage (Connor et al., 1994CONNOR, R.J., KAWAOKA, Y., WEBSTER, R.G. and PAULSON, J.C., 1994. Receptor specificity in human, avian, and equine H2 and H3 influenza virus isolates. Virology, vol. 205, no. 1, pp. 17-23. http://dx.doi.org/10.1006/viro.1994.1615. PMid:7975212.
http://dx.doi.org/10.1006/viro.1994.1615...
). Similar linkage pattern is recognised by the equine influenza virus found on the cells lining of the equine upper respiratory tract (Suzuki et al., 2000SUZUKI, Y., ITO, T., SUZUKI, T., HOLLAND JUNIOR, R.E., CHAMBERS, T.M., KISO, M., ISHIDA, H. and KAWAOKA, Y., 2000. Sialic acid species as a determinant of the host range of influenza A viruses. Journal of Virology, vol. 74, no. 24, pp. 11825-11831. http://dx.doi.org/10.1128/JVI.74.24.11825-11831.2000. PMid:11090182.
http://dx.doi.org/10.1128/JVI.74.24.1182...
). In contrast the influenza viruses found in humans binds with SA 2, 6 receptors and these receptors predominate in their respiratory tract (Couceiro et al., 1993COUCEIRO, J.N.S., PAULSON, J.C. and BAUM, L.G., 1993. Influenza virus strains selectively recognize sialyloligosaccharides on human respiratory epithelium; the role of the host cell in selection of hemagglutinin receptor specificity. Virus Research, vol. 29, no. 2, pp. 155-165. http://dx.doi.org/10.1016/0168-1702(93)90056-S. PMid:8212857.
http://dx.doi.org/10.1016/0168-1702(93)9...
). The SA 2, 3 predominance linkage preferred by the equine influenza viruses on the canine respiratory tracheal cells has also been demonstrated (Daly et al., 2008DALY, J.M., BLUNDEN, A.S., MACRAE, S., MILLER, J., BOWMAN, S.J., KOLODZIEJEK, J., NOWOTNY, N. and SMITH, K.C., 2008. Transmission of equine influenza virus to English foxhounds. Emerging Infectious Diseases, vol. 14, no. 3, pp. 461-464. http://dx.doi.org/10.3201/eid1403.070643. PMid:18325262.
http://dx.doi.org/10.3201/eid1403.070643...
). This availability of the relevant receptors for EIVs on the canine respiratory tract poses a question of why the equine influenza infection in dogs had not been reported before 2005. However multiple reports of EIVs transmission into dogs have recently published (Newton et al., 2007NEWTON, R., COOKE, A., ELTON, D., BRYANT, N., RASH, A., BOWMAN, S., BLUNDEN, T., MILLER, J., HAMMOND, T.-A., CAMM, I. and DAY, M., 2007. Canine influenza virus: cross-species transmission from horses. The Veterinary Record, vol. 161, no. 4, pp. 142-143. http://dx.doi.org/10.1136/vr.161.4.142-a. PMid:17660470.
http://dx.doi.org/10.1136/vr.161.4.142-a...
) suggested that EIVs recently circulating in the equines are particularly transmissible.
The phenomena of transmission possibility depend upon the interaction nature between two species. Although the transmission occurs through aerosol route between man-to-man, horse-to-horse and it could also occur direct contact (Lowen and Palese, 2009LOWEN, A. and PALESE, P., 2009. Transmission of influenza virus in temperate zones is predominantly by aerosol, in the tropics by contact: a hypothesis. PLoS Currents, vol. 1, p. RRN1002. http://dx.doi.org/10.1371/currents.RRN1002. PMid:20025197.
http://dx.doi.org/10.1371/currents.RRN10...
). It has also been reported that EI can also be transmitted from experimentally infected equine to canines housed in the same premises (Yamanaka et al., 2009YAMANAKA, T., NEMOTO, M., TSUJIMURA, K., KONDO, T. and MATSUMURA, T., 2009. Interspecies transmission of equine influenza virus (H3N8) to dogs by close contact with experimentally infected horses. Veterinary Microbiology, vol. 139, no. 3-4, pp. 351-355. http://dx.doi.org/10.1016/j.vetmic.2009.06.015. PMid:19596528.
http://dx.doi.org/10.1016/j.vetmic.2009....
). It was confirmed by the reports by (Krueger et al., 2014KRUEGER, W.S., HEIL, G.L., YOON, K.J. and GRAY, G.C., 2014. No evidence for zoonotic transmission of H 3 N 8 canine influenza virus among US adults occupationally exposed to dogs. Influenza and Other Respiratory Viruses, vol. 8, no. 1, pp. 99-106. http://dx.doi.org/10.1111/irv.12208. PMid:24237615.
http://dx.doi.org/10.1111/irv.12208...
, Yondon et al., 2014YONDON, M., ZAYAT, B., NELSON, M.I., HEIL, G.L., ANDERSON, B.D., LIN, X., HALPIN, R.A., MCKENZIE, P.P., WHITE, S.K., WENTWORTH, D.E. and GRAY, G.C., 2014. Equine influenza A (H3N8) virus isolated from Bactrian camel, Mongolia. Emerging Infectious Diseases, vol. 20, no. 12, pp. 2144-2147. http://dx.doi.org/10.3201/eid2012.140435. PMid:25418532.
http://dx.doi.org/10.3201/eid2012.140435...
); that dogs got EI being kept near to the infected equines though no evidence of transmission could be established. Further evidence has been reported that equines may not be the only dead-end-hosts; it has been confirmed by the characterization of the two isolated viral strains from pigs showing the respiratory infection in 2005-2006 (Tu et al., 2009TU, J., ZHOU, H., JIANG, T., LI, C., ZHANG, A., GUO, X., ZOU, W., CHEN, H. and JIN, M., 2009. Isolation and molecular characterization of equine H3N8 influenza viruses from pigs in China. Archives of Virology, vol. 154, no. 5, pp. 887-890. http://dx.doi.org/10.1007/s00705-009-0381-1. PMid:19396578.
http://dx.doi.org/10.1007/s00705-009-038...
).
Recently, two different lineages H17N10 and H18N11 of influenza viruses have been identified in bats. This discovery has provided a novel insight into evolution and origin of influenza-A viruses beyond hypothesis of waterfowls or shorebirds predominantly common as primary natural reservoirs. It is also known that H3 HA is having broad pathogenic potential but analysis of this gene of influenza-A virus suggests that canine and equine H3 have been evolved independently to the H3 avian, human and the swine flu viruses (Shi et al., 2010SHI, W., LEI, F., ZHU, C., SIEVERS, F. and HIGGINS, D.G., 2010. A complete analysis of HA and NA genes of influenza A viruses. PLoS One, vol. 5, no. 12, pp. e14454. http://dx.doi.org/10.1371/journal.pone.0014454. PMid:21209922.
http://dx.doi.org/10.1371/journal.pone.0...
). As mentioned earlier that a correlation between the receptor binding sites and host specificity for EIVs exists. On the other side the human adapted influenza viruses mostly recognize and binds SA 2, 6 receptors on host cells (Daly et al., 2011DALY, J.M., MACRAE, S., NEWTON, J.R., WATTRANG, E. and ELTON, D.M., 2011. Equine influenza: a review of an unpredictable virus. Veterinary Journal, vol. 189, no. 1, pp. 7-14. http://dx.doi.org/10.1016/j.tvjl.2010.06.026. PMid:20685140.
http://dx.doi.org/10.1016/j.tvjl.2010.06...
). Virus shedding and seroconversions have also recorded in the human volunteers being inoculated with EIVs (Kasel and Couch, 1969KASEL, J.A. and COUCH, R., 1969. Experimental infections in man and horses with influenza A viruses. Bulletin of the World Health Organization, vol. 41, no. 3, pp. 447-452. PMid:5309454.) but it is still not understood if potential for such kind of transmission exists. But it is also possible that new influenza subtypes emerges in equines from avian reservoirs without replacing the currently circulating H3N8 virus (Raiffa et al., 1999RAIFFA, H., HAMMOND JOHN, S. and KEENEY RALPH, L., 1999. Smart choices: a practical guide to making better life decisions. Boston: Harvard Business School Press.). Cross species transmission of the avian H3N8 viruses into equines in china in 1989 was most closely related to AIVs than other EIVs (Guo et al., 1992GUO, Y., WANG, M., KAWAOKA, Y., GORMAN, O., ITO, T., SAITO, T. and WEBSTER, R.G., 1992. Characterization of a new avian-like influenza A virus from horses in China. Virology, vol. 188, no. 1, pp. 245-255. http://dx.doi.org/10.1016/0042-6822(92)90754-D. PMid:1314452.
http://dx.doi.org/10.1016/0042-6822(92)9...
). In another study H5N1 of avian origin has been isolated from donkeys in Egypt (Abdel-Moneim et al., 2010ABDEL-MONEIM, A.S., ABDEL-GHANY, A.E. and SHANY, S.A., 2010. Isolation and characterization of highly pathogenic avian influenza virus subtype H5N1 from donkeys. Journal of Biomedical Science, vol. 17, no. 1, pp. 25. http://dx.doi.org/10.1186/1423-0127-17-25. PMid:20398268.
http://dx.doi.org/10.1186/1423-0127-17-2...
) describing a highly contagious AIV as equine infectious agent, also raising the questions about role of the donkeys in H5N1 spread to birds, equines and Humans. In another recent study in Pakistan avian origin viruses have been isolated and characterized in equines (Crawford et al., 2005CRAWFORD, P.C., DUBOVI, E.J., CASTLEMAN, W.L., STEPHENSON, I., GIBBS, E., CHEN, L., SMITH, C., HILL, R.C., FERRO, P., POMPEY, J., BRIGHT, R.A., MEDINA, M.J., JOHNSON, C.M., OLSEN, C.W., COX, N.J., KLIMOV, A.I., KATZ, J.M. and DONIS, R.O., 2005. Transmission of equine influenza virus to dogs. Science, vol. 310, no. 5747, pp. 482-485. http://dx.doi.org/10.1126/science.1117950. PMid:16186182.
http://dx.doi.org/10.1126/science.111795...
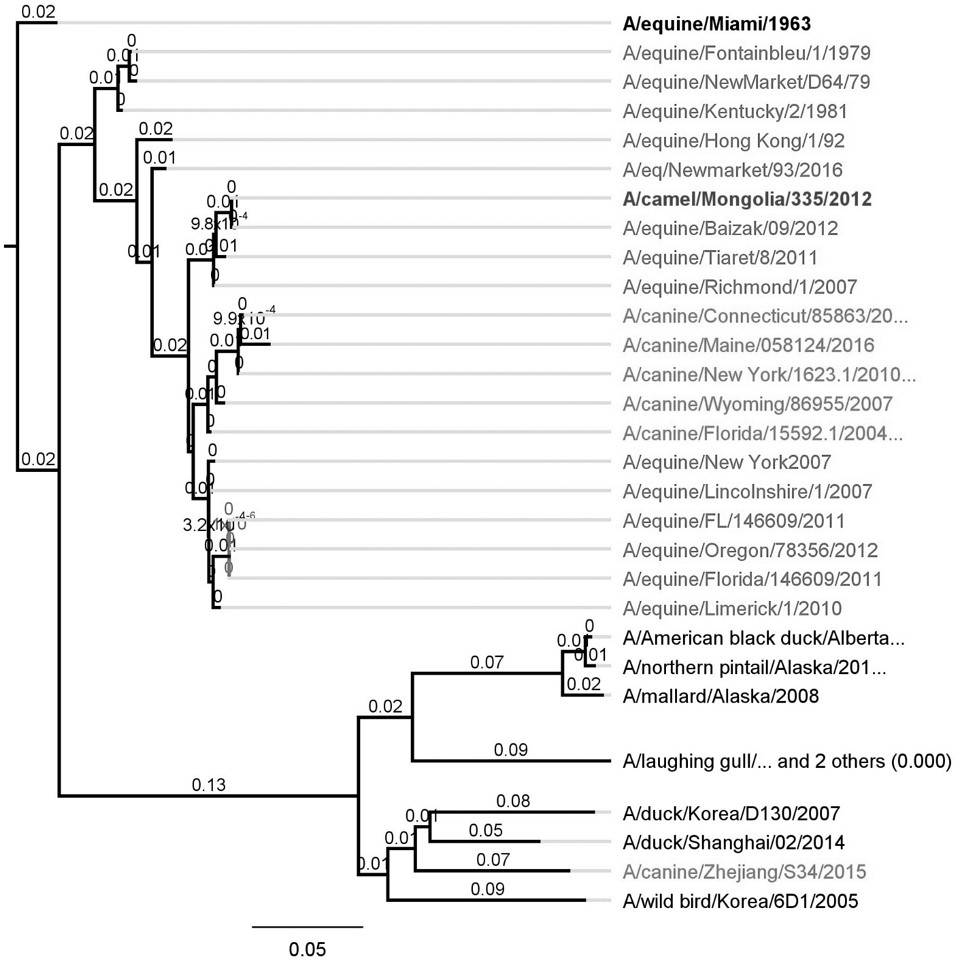
). It is the most recent findings highlighting the emerging threat of avian virus’s transmission into equines and high rate of mutation. Phylogenetic analysis of H3N8 isolated from different species is given in Figure 2.
Phylogenetic analysis of EIV H3N8 based on HA gene amino-acid isolated in different species globally.
3.1.5. Spread and transmission of equine influenza viruses
EI is primarily a seasonal infection of equines usually occurs in the form of epidemic, mostly rampant in waves, followed by a relative calm period. EIV is a contagious disease in nature and spreads by respiratory route by direct contact of infectious and susceptible equines in very close proximity. In susceptible and unvaccinated equines the short incubation duration and the persistent coughing releases a large amount of viruses into immediate environment that contributes highly to the rapid spread and transmission of the infection (King and Macdonald, 2004KING, E. and MACDONALD, D., 2004. Report of the Board of Inquiry appointed by the Board of the National Horseracing Authority to conduct enquiry into the causes of the equine influenza which started in the Western cape in early December 2003 and spread to the Eastern Cape and Gauteng. Australian Equine Veterinarian, vol. 23, pp. 139-142., Guthrie et al., 1999GUTHRIE, A., STEVENS, K. and BOSMAN, P., 1999. The circumstances surrounding the outbreak and spread of equine influenza in South Africa (emergency management). In: Office international des épizooties, ed. Revue Scientifique et Technique de l'OIE. Paris: Office international des épizooties.). Severe outbreaks takes affects mostly unvaccinated equine population or when a new viral strain infects vaccinated equines. In susceptible equines morbidity rate could reach to 100%. The global EIV distribution is associated with movement of equines for participation in different sports competitions or breeding purpose. In an outbreak recently in Uruguay in 2012, 2000 horses were affected by EIVs due to this race meetings were post-ponded for several weeks and equine movements out of the country was banned (Beuttemmüller et al., 2016BEUTTEMMÜLLER, E.A., WOODWARD, A., RASH, A., FERRAZ, L.E.S., ALFIERI, A.F., ALFIERI, A.A. and ELTON, D., 2016. Characterisation of the epidemic strain of H3N8 equine influenza virus responsible for outbreaks in South America in 2012. Virology Journal, vol. 13, no. 1, pp. 45. http://dx.doi.org/10.1186/s12985-016-0503-9. PMid:26993620.
http://dx.doi.org/10.1186/s12985-016-050...
).
3.1.6. Recently reported outbreaks
Due to regular mutation of the virus extensive use of the vaccines in several horse populations epidemics of EI continue to occur. In Newmarket, UK in 2003 a widespread outbreak occurred in vaccinated population (Newton et al., 2006NEWTON, J.R, DALY, J., SPENCER, L. and MUMFORD, J., 2006. Description of the outbreak of equine influenza (H3N8) in the United Kingdom in 2003, during which recently vaccinated horses in Newmarket developed respiratory disease. The Veterinary Record, vol. 158, no. 6, pp. 185-192. http://dx.doi.org/10.1136/vr.158.6.185. PMid:16474051.
http://dx.doi.org/10.1136/vr.158.6.185...
, Barquero et al., 2007BARQUERO, N., DALY, J.M. and NEWTON, J.R., 2007. Risk factors for influenza infection in vaccinated racehorses: lessons from an outbreak in Newmarket, UK in 2003. Vaccine, vol. 25, no. 43, pp. 7520-7529. http://dx.doi.org/10.1016/j.vaccine.2007.08.038. PMid:17889409.
http://dx.doi.org/10.1016/j.vaccine.2007...
). In 2003-2004 South Africa faced a second major EI outbreak (Guthrie, 2006GUTHRIE, A., 2006. Equine influenza in South Africa, 2003 outbreak. In: Proceedings of the Proceedings of the 9th International Congress of World Equine Veterinary Association, 22-26 January 2006, Marrakech, Morocco. Marrakech: World Equine Veterinary Association.). In 2008 in India an outbreak of EI after an interval of almost 20 years was reported (Virmani et al., 2010VIRMANI, N., BERA, B., SINGH, B., SHANMUGASUNDARAM, K., GULATI, B., BARUA, S., VAID, R., GUPTA, A. and SINGH, R., 2010. Equine influenza outbreak in India (2008–09): virus isolation, sero-epidemiology and phylogenetic analysis of HA gene. Veterinary Microbiology, vol. 143, no. 2-4, pp. 224-237. http://dx.doi.org/10.1016/j.vetmic.2009.12.007. PMid:20053509.
http://dx.doi.org/10.1016/j.vetmic.2009....
). Australia which was considered free of EIV previously also experienced a severe outbreak of EI in 2007 (Alder, 2008ALDER, M., ed., 2008. Summary of the Australian equine influenza outbreak. The Veterinary Record, vol. 163, no. 13, pp. 378. http://dx.doi.org/10.1136/vr.163.13.378. PMid:19031639.
http://dx.doi.org/10.1136/vr.163.13.378...
, Daly et al., 2011DALY, J.M., MACRAE, S., NEWTON, J.R., WATTRANG, E. and ELTON, D.M., 2011. Equine influenza: a review of an unpredictable virus. Veterinary Journal, vol. 189, no. 1, pp. 7-14. http://dx.doi.org/10.1016/j.tvjl.2010.06.026. PMid:20685140.
http://dx.doi.org/10.1016/j.tvjl.2010.06...
). A recent outbreak was reported from Pakistan infecting donkeys and horses in the northern parts of Pakistan bordering Afghanistan; where the index case was reported being sourced from Afghanistan by importing horses and donkeys across the border (Arthur, 2014ARTHUR, M., 2014. Institute for health metrics and evaluation. Nursing Standard, vol. 28, no. 42, pp. 32. http://dx.doi.org/10.7748/ns.28.42.32.s33. PMid:25005408.
http://dx.doi.org/10.7748/ns.28.42.32.s3...
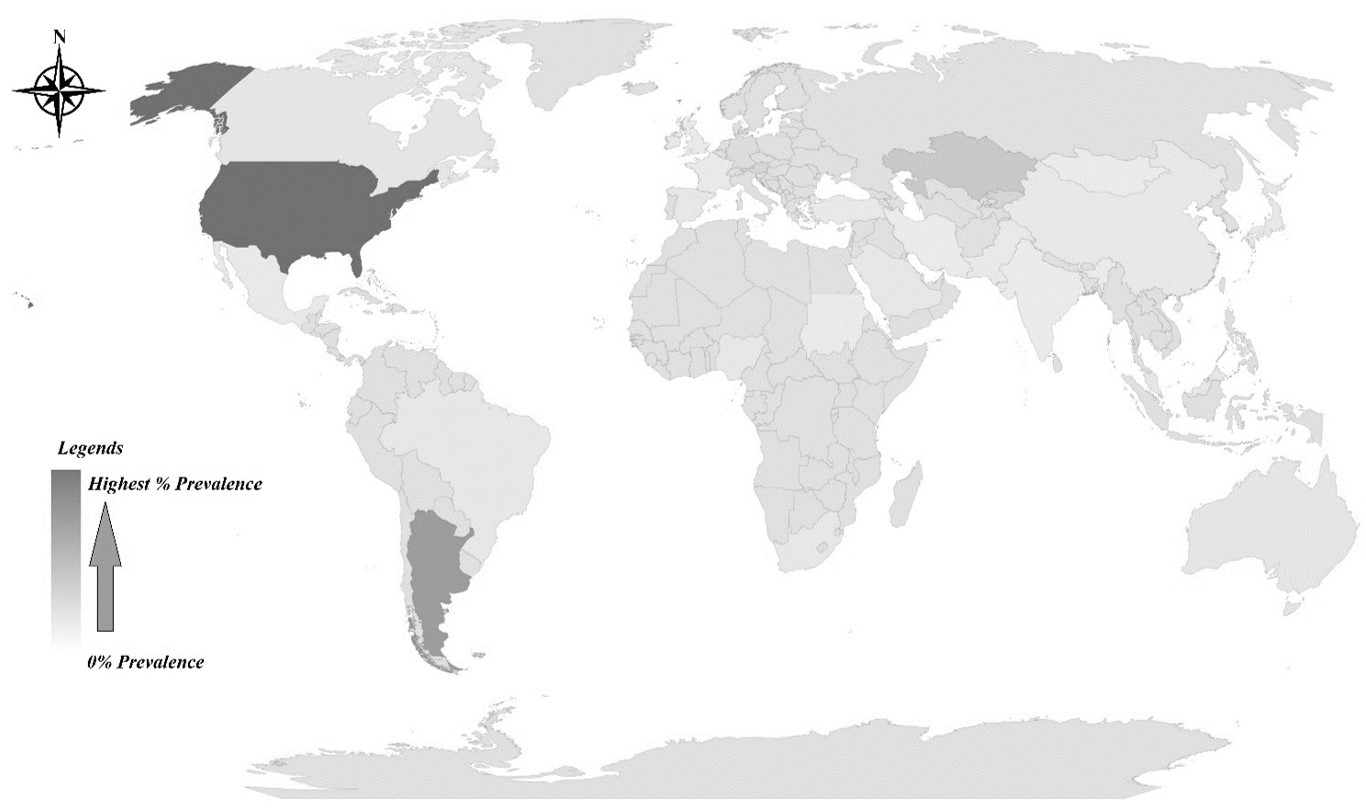
). Analyzing Prevalence and several outbreaks data from several articles from different regions globally is concluded here (Figure 3).
Geographical representation (based on literature review) of EIV outbreak severity globally based on the no of cases reported.
3.1.7. Molecular epidemiology and evolution of EIV’s
The molecular epidemiology of EIVs is quite different from influenza viruses those have established in avian, aquatic species and human beings. Historically, phylogenetic analysis of the HA gene sequences of EIVs H3N8 revealed that it had been evolved as single lineage for two decades at least (Kawaoka et al., 1989KAWAOKA, Y., BEAN, W.J. and WEBSTER, R.G., 1989. Evolution of the hemagglutinin of equine H3 influenza viruses. Virology, vol. 169, no. 2, pp. 283-292. http://dx.doi.org/10.1016/0042-6822(89)90153-0. PMid:2705299.
http://dx.doi.org/10.1016/0042-6822(89)9...
), which later on diverged into couple of distinct lineage in 1980s (Daly et al., 1996DALY, J.M., LAI, A., BINNS, M., CHAMBERS, T., BARRANDEGUY, M. and MUMFORD, J., 1996. Antigenic and genetic evolution of equine H3N8 influenza A viruses. The Journal of General Virology, vol. 77, no. Pt 4, pp. 661-671. http://dx.doi.org/10.1099/0022-1317-77-4-661. PMid:8627254.
http://dx.doi.org/10.1099/0022-1317-77-4...
). Viruses of one lineage were mostly isolated from equines of American continent, while the other lineage comprised the viruses exclusively isolated from equines of European and Asian countries. A recent phylogenetic tree of H3N8 EIVs strain constructed here is very complex (Figure 4).
Phylogenetic analysis based on nucleotide sequence of HA-gene EIV H3N8-viruses different clades isolated in different regions of around the world.
The American lineage EIV viral strains currently dominate and have evolved into three different clades. Within American lineage a variant sub lineage known as Florida lineage caused an outbreak in 2003 in Newmarket and spread across the European countries thus isolates being identified in Europe since 2003 belongs to this lineage (Damiani et al., 2008DAMIANI, A.M., SCICLUNA, M., CIABATTI, I., CARDETI, G., SALA, M., VULCANO, G., CORDIOLI, P., MARTELLA, V., AMADDEO, D. and AUTORINO, G., 2008. Genetic characterization of equine influenza viruses isolated in Italy between 1999 and 2005. Virus Research, vol. 131, no. 1, pp. 100-105. http://dx.doi.org/10.1016/j.virusres.2007.08.001. PMid:17889395.
http://dx.doi.org/10.1016/j.virusres.200...
, Bryant et al., 2009BRYANT, N.A., RASH, A.S., RUSSELL, C.A., ROSS, J., COOKE, A., BOWMAN, S., MACRAE, S., LEWIS, N.S., PAILLOT, R., ZANONI, R., MEIER, H., GRIFFITHS, L.A., DALY, J.M., TIWARI, A., CHAMBERS, T.M., NEWTON, J.R. and ELTON, D.M., 2009. Antigenic and genetic variations in European and North American equine influenza virus strains (H3N8) isolated from 2006 to 2007. Veterinary Microbiology, vol. 138, no. 1-2, pp. 41-52. http://dx.doi.org/10.1016/j.vetmic.2009.03.004. PMid:19346084.
http://dx.doi.org/10.1016/j.vetmic.2009....
, Rozek et al., 2009ROZEK, W., PURZYCKA, M., POLAK, M.P., GRADZKI, Z. and ZMUDZINSKI, J.F., 2009. Genetic typing of equine influenza virus isolated in Poland in 2005 and 2006. Virus Research, vol. 145, no. 1, pp. 121-126. http://dx.doi.org/10.1016/j.virusres.2009.06.017. PMid:19580828.
http://dx.doi.org/10.1016/j.virusres.200...
). However the two clades could be differentiated in this lineage; the Florida Clade 1 been isolated the North American region since 2003 (e.g., Ohio/2003) and Florida Clade 2 isolated from Europe (e.g., Newmarket/5/03). Florida Clade 2 EIVs caused major outbreaks in China, India and Mongolia in 2007-2009 [51, 59]. While Florida Clade-1 was responsible for an EI epidemic in South Africa in 2003 and in Australia and Japan in 2007 (Bryant et al., 2009BRYANT, N.A., RASH, A.S., RUSSELL, C.A., ROSS, J., COOKE, A., BOWMAN, S., MACRAE, S., LEWIS, N.S., PAILLOT, R., ZANONI, R., MEIER, H., GRIFFITHS, L.A., DALY, J.M., TIWARI, A., CHAMBERS, T.M., NEWTON, J.R. and ELTON, D.M., 2009. Antigenic and genetic variations in European and North American equine influenza virus strains (H3N8) isolated from 2006 to 2007. Veterinary Microbiology, vol. 138, no. 1-2, pp. 41-52. http://dx.doi.org/10.1016/j.vetmic.2009.03.004. PMid:19346084.
http://dx.doi.org/10.1016/j.vetmic.2009....
). The phylogenetic analysis points to the sporadic transmission of EIVs from North America into the Europe and other regions of the World, evolution and mutation have happened in a more linear nature around 1993 to 2003, followed by a more rapid and localized divergent evolution of the EIVs (Figure 5).
The recent reports specifically from Pakistan highlights the future threats of the virus transmission between the equines, wild birds and human beings. As the farming practices and close contact of different mammalian species with domestic and wild birds, and also the close contact of farmers to their animals highly supports the interspecies transmission (Khan et al., 2018KHAN, A., MUSHTAQ, M.H., AHMAD, M.U.D., NAZIR, J., FATIMA, Z., KHAN, A. and FAROOQI, S.H., 2018. Molecular epidemiology of the two internal genes of equine influenza H3N8 virus isolated in Pakistan 2015-16. Pakistan Veterinary Journal, vol. 38, no. 1, pp. 96-100.). By analogy, such mutations are considered a type of optimal EIVs adaptation with the lineage. Like human and swine influenza lineages, the EIV lineage shows a more continuous evolutionary advancement with time. It’s important to note the mutation rate of mammalian influenza viruses’ nucleoprotein is much greater than the AIVs. Amino acid alignment of H3N8 EIV isolated in different species have been aligned to assess the mutation and genetic variation (Figure 6) among the isolates at specie level.
3.2. Evidences of EIV zoonosis
3.2.1. Historical evidence
Historically there are several studies that had been reported evidence of EIV infections in human beings. Here we analyzed several articles data and we concluded considerable evidence of EIV zoonotic history in man. Although some of the scientists explained confounders carefully (Legrand et al., 2013LEGRAND, L.J, PITEL, P.H., MARCILLAUD‐PITEL, C., CULLINANE, A., COUROUCE, A., FORTIER, G., FREYMUTH, F. and PRONOST, S., 2013. Surveillance of equine influenza viruses through the RESPE network in France from November 2005 to October 2010. Equine Veterinary Journal, vol. 45, no. 6, pp. 776-783. http://dx.doi.org/10.1111/evj.12100. PMid:23662725.
http://dx.doi.org/10.1111/evj.12100...
) and reported several observations. The primitive observation was from 1658 to 20th century outbreaks in equine population from EIVs often took place couple of weeks prior to human influenza infections. More specifically, most of the scientists believe that 1889 pandemic in humans was caused most probably by H3N8 EIVs (Legrand et al., 2013LEGRAND, L.J, PITEL, P.H., MARCILLAUD‐PITEL, C., CULLINANE, A., COUROUCE, A., FORTIER, G., FREYMUTH, F. and PRONOST, S., 2013. Surveillance of equine influenza viruses through the RESPE network in France from November 2005 to October 2010. Equine Veterinary Journal, vol. 45, no. 6, pp. 776-783. http://dx.doi.org/10.1111/evj.12100. PMid:23662725.
http://dx.doi.org/10.1111/evj.12100...
, Back et al., 2016BACK, H., BERNDTSSON, L.T., GRÖNDAHL, G., STÅHL, K., PRINGLE, J. and ZOHARI, S., 2016. The first reported Florida clade 1 virus in the Nordic countries, isolated from a Swedish outbreak of equine influenza in 2011. Veterinary Microbiology, vol. 184, pp. 1-6. http://dx.doi.org/10.1016/j.vetmic.2015.12.010. PMid:26854337.
http://dx.doi.org/10.1016/j.vetmic.2015....
). Several serological studies also reported antibodies against EIV H3N8 in people lived in the 1892 era (Bryant et al., 2009BRYANT, N.A., RASH, A.S., RUSSELL, C.A., ROSS, J., COOKE, A., BOWMAN, S., MACRAE, S., LEWIS, N.S., PAILLOT, R., ZANONI, R., MEIER, H., GRIFFITHS, L.A., DALY, J.M., TIWARI, A., CHAMBERS, T.M., NEWTON, J.R. and ELTON, D.M., 2009. Antigenic and genetic variations in European and North American equine influenza virus strains (H3N8) isolated from 2006 to 2007. Veterinary Microbiology, vol. 138, no. 1-2, pp. 41-52. http://dx.doi.org/10.1016/j.vetmic.2009.03.004. PMid:19346084.
http://dx.doi.org/10.1016/j.vetmic.2009....
, Garner et al., 2011GARNER, M.G., COWLED, B., EAST, I., MOLONEY, B. and KUNG, N., 2011. Evaluating the effectiveness of early vaccination in the control and eradication of equine influenza: a modelling approach. Preventive Veterinary Medicine, vol. 99, no. 1, pp. 15-27. http://dx.doi.org/10.1016/j.prevetmed.2010.02.007. PMid:20236718.
http://dx.doi.org/10.1016/j.prevetmed.20...
). Isolation of avian origin influenza viruses from equine population (Khan et al., 2018KHAN, A., MUSHTAQ, M.H., AHMAD, M.U.D., NAZIR, J., FATIMA, Z., KHAN, A. and FAROOQI, S.H., 2018. Molecular epidemiology of the two internal genes of equine influenza H3N8 virus isolated in Pakistan 2015-16. Pakistan Veterinary Journal, vol. 38, no. 1, pp. 96-100.) in Pakistan highlights the chances of influenza virus transmission between horses and man (Table 1). Other studies reporting the evidence of H3N8 zoonosis possibility is given in Table 1.
3.2.2. Experimental evidence
In early 1960s challenge experimental studies on human with EIV were conducted and another experimental challenge study conducted on volunteers working in hospital with H3N8 live equine virus (A/Equi-2/Miami/63). These participants after challenge inoculation were carefully observed (Kasel et al., 1965KASEL, J.A., ALFORD, R.H., KNIGHT, V., WADDELL, G.H. and SIGEL, M.M., 1965. Experimental infection of human volunteers with equine influenza virus. Nature, vol. 206, no. 4979, pp. 41-43. http://dx.doi.org/10.1038/206041a0. PMid:14334358.
http://dx.doi.org/10.1038/206041a0...
). After 3 days viable EIV virus was isolated but only a single participant developed clinical signs. But in another study conducted in 1966; challenging human volunteer with H3N8 EIV (Alford et al., 1967ALFORD, R.H., KASEL, J.A., LEHRICH, J.R. and KNIGHT, V., 1967. Human responses to experimental infection with influenza A/Equi 2 virus. American Journal of Epidemiology, vol. 86, no. 1, pp. 185-192. http://dx.doi.org/10.1093/oxfordjournals.aje.a120723. PMid:4951557.
http://dx.doi.org/10.1093/oxfordjournals...
) several volunteers were observed who developed proper clinical signs and virus was isolated after 3 days of challenge. While in another challenge study of EIV H3N8 in humans in 1969 (Kasel and Couch, 1969KASEL, J.A. and COUCH, R., 1969. Experimental infections in man and horses with influenza A viruses. Bulletin of the World Health Organization, vol. 41, no. 3, pp. 447-452. PMid:5309454., Couch et al., 1969COUCH, R., DOUGLAS, R., KASEL, J., RIGGS, S. and KNIGHT, V., 1969. Production of the influenza syndrome in man with equine influenza virus. Nature, vol. 224, no. 5218, pp. 512-514. http://dx.doi.org/10.1038/224512a0.
http://dx.doi.org/10.1038/224512a0...
); several patients developed signs of clinical illness (Su et al., 2014SU, S., WANG, L., FU, X., HE, S., HONG, M., ZHOU, P., LAI, A., GRAY, G. and LI, S., 2014. Equine influenza A (H3N8) virus infection in cats. Emerging Infectious Diseases, vol. 20, no. 12, pp. 2096-2099. http://dx.doi.org/10.3201/eid2012.140867. PMid:25417790.
http://dx.doi.org/10.3201/eid2012.140867...
). After passaging the virus isolated form the challenged participants after challenge showed that virus H3N8 was not attenuated after being passaged in humans, as it was found still capable of infecting and causing illness in equines.
3.2.3. Natural evidence of EIV infection in humans
Several reports and historical scientific drafts had been published, reporting that horse’s humans, dogs and cats concomitantly developed influenza like infections (Yondon et al., 2013YONDON, M., HEIL, G.L., BURKS, J.P., ZAYAT, B., WALTZEK, T.B., JAMIYAN, B.O., MCKENZIE, P.P., KRUEGER, W.S., FRIARY, J.A. and GRAY, G.C., 2013. Isolation and characterization of H 3 N 8 equine influenza A virus associated with the 2011 epizootic in M ongolia. Influenza and Other Respiratory Viruses, vol. 7, no. 5, pp. 659-665. http://dx.doi.org/10.1111/irv.12069. PMid:23289427.
http://dx.doi.org/10.1111/irv.12069...
, He, 2012HE, Q., 2012. Isolation and whole genome sequence analysis of equine H9N2 influenza virus in Guangxi Province, China. Nanning, China: Guangxi University. Masters Dissertation., Landolt, 2014LANDOLT, G.A., 2014. Equine influenza virus. Veterinary Clinics of North America: Equine Practice, vol. 30, no. 3, pp. 507-522. PMid:25282321.). The highest man to horse population ration is found in Mongolia globally and have usually faced large number EIV epizootics (Crispe et al., 2011CRISPE, E., FINLAISON, D., HURT, A. and KIRKLAND, P., 2011. Infection of dogs with equine influenza virus: evidence for transmission from horses during the Australian outbreak. Australian Veterinary Journal, vol. 89, suppl. 1, pp. 27-28. http://dx.doi.org/10.1111/j.1751-0813.2011.00734.x. PMid:21711279.
http://dx.doi.org/10.1111/j.1751-0813.20...
). Some reports suggest that Mongolian children’s developed respiratory illnesses post EIV infected horse’s exposure (Khurelbaatar et al., 2013KHURELBAATAR, N., KRUEGER, W.S., HEIL, G.L., DARMAA, B., ULZIIMAA, D., TSERENNOROV, D., BATERDENE, A., ANDERSON, B.D. and GRAY, G.C., 2013. Sparse evidence for equine or avian influenza virus infections among Mongolian adults with animal exposures. Influenza and Other Respiratory Viruses, vol. 7, no. 6, pp. 1246-1250. http://dx.doi.org/10.1111/irv.12148. PMid:23941547.
http://dx.doi.org/10.1111/irv.12148...
). In Australia a cross sectional study was conducted following the 2007 outbreak on human beings exposed to the infected horses. Serum samples were collected from these individuals for the presence of antibodies against the H3N8 EIV. Only few participants were found with low antibodies titers which might also be associated with cross reacting antibodies from vaccines or influenza viruses from humans (Larson et al., 2015LARSON, K.R.L., HEIL, G.L., CHAMBERS, T.M., CAPUANO, A., WHITE, S.K. and GRAY, G.C., 2015. Serological evidence of equine influenza infections among persons with horse exposure, Iowa. Journal of Clinical Virology, vol. 67, pp. 78-83. http://dx.doi.org/10.1016/j.jcv.2015.04.009. PMid:25959164.
http://dx.doi.org/10.1016/j.jcv.2015.04....
).
4. Discussion
Equine influenza viruses (EIVs) have been isolated from different mammalian species. Several experimental challenge studies in humans have reported H3N8 EIV; clearly documenting the possibility of H3N8 infection in humans, though the signs may not exist (Kasel et al., 1965KASEL, J.A., ALFORD, R.H., KNIGHT, V., WADDELL, G.H. and SIGEL, M.M., 1965. Experimental infection of human volunteers with equine influenza virus. Nature, vol. 206, no. 4979, pp. 41-43. http://dx.doi.org/10.1038/206041a0. PMid:14334358.
http://dx.doi.org/10.1038/206041a0...
, Alford et al., 1967ALFORD, R.H., KASEL, J.A., LEHRICH, J.R. and KNIGHT, V., 1967. Human responses to experimental infection with influenza A/Equi 2 virus. American Journal of Epidemiology, vol. 86, no. 1, pp. 185-192. http://dx.doi.org/10.1093/oxfordjournals.aje.a120723. PMid:4951557.
http://dx.doi.org/10.1093/oxfordjournals...
, Kasel and Couch, 1969KASEL, J.A. and COUCH, R., 1969. Experimental infections in man and horses with influenza A viruses. Bulletin of the World Health Organization, vol. 41, no. 3, pp. 447-452. PMid:5309454.). Amongst them the most important was that of 1960s observation to report here with no attenuation real evidence. The 2005 observation study in horse exposed humans in Iowa was the most compelling evidence for human infection with the EIV viruses (Larson et al., 2015LARSON, K.R.L., HEIL, G.L., CHAMBERS, T.M., CAPUANO, A., WHITE, S.K. and GRAY, G.C., 2015. Serological evidence of equine influenza infections among persons with horse exposure, Iowa. Journal of Clinical Virology, vol. 67, pp. 78-83. http://dx.doi.org/10.1016/j.jcv.2015.04.009. PMid:25959164.
http://dx.doi.org/10.1016/j.jcv.2015.04....
) Searching the Influenza Research Data base (IRD), it was found that segments associated to influenza A H5N1, H1N8, H7N1 and H9N2 strains been detected among the samples which were collected from the equines. It is also an established fact that equines are susceptible to some diverse strains of influenza type A viruses than believed previously. For example recently a novel virus was isolated in Pakistan from Equines (Khan et al., 2018KHAN, A., MUSHTAQ, M.H., AHMAD, M.U.D., NAZIR, J., FATIMA, Z., KHAN, A. and FAROOQI, S.H., 2018. Molecular epidemiology of the two internal genes of equine influenza H3N8 virus isolated in Pakistan 2015-16. Pakistan Veterinary Journal, vol. 38, no. 1, pp. 96-100.) and in 2011 H9N2 viruses was also detected and isolated from horses in China. It’s also an interesting finding that for H3N8 EIV viruses horses are not always dead end hosts. It has also been isolated in dogs in UK, USA and Australia (Crawford et al., 2005CRAWFORD, P.C., DUBOVI, E.J., CASTLEMAN, W.L., STEPHENSON, I., GIBBS, E., CHEN, L., SMITH, C., HILL, R.C., FERRO, P., POMPEY, J., BRIGHT, R.A., MEDINA, M.J., JOHNSON, C.M., OLSEN, C.W., COX, N.J., KLIMOV, A.I., KATZ, J.M. and DONIS, R.O., 2005. Transmission of equine influenza virus to dogs. Science, vol. 310, no. 5747, pp. 482-485. http://dx.doi.org/10.1126/science.1117950. PMid:16186182.
http://dx.doi.org/10.1126/science.111795...
, Yamanaka et al., 2009YAMANAKA, T., NEMOTO, M., TSUJIMURA, K., KONDO, T. and MATSUMURA, T., 2009. Interspecies transmission of equine influenza virus (H3N8) to dogs by close contact with experimentally infected horses. Veterinary Microbiology, vol. 139, no. 3-4, pp. 351-355. http://dx.doi.org/10.1016/j.vetmic.2009.06.015. PMid:19596528.
http://dx.doi.org/10.1016/j.vetmic.2009....
), in cats and China (Tu et al., 2009TU, J., ZHOU, H., JIANG, T., LI, C., ZHANG, A., GUO, X., ZOU, W., CHEN, H. and JIN, M., 2009. Isolation and molecular characterization of equine H3N8 influenza viruses from pigs in China. Archives of Virology, vol. 154, no. 5, pp. 887-890. http://dx.doi.org/10.1007/s00705-009-0381-1. PMid:19396578.
http://dx.doi.org/10.1007/s00705-009-038...
) and in camels from Mongolia (Yondon et al., 2013YONDON, M., HEIL, G.L., BURKS, J.P., ZAYAT, B., WALTZEK, T.B., JAMIYAN, B.O., MCKENZIE, P.P., KRUEGER, W.S., FRIARY, J.A. and GRAY, G.C., 2013. Isolation and characterization of H 3 N 8 equine influenza A virus associated with the 2011 epizootic in M ongolia. Influenza and Other Respiratory Viruses, vol. 7, no. 5, pp. 659-665. http://dx.doi.org/10.1111/irv.12069. PMid:23289427.
http://dx.doi.org/10.1111/irv.12069...
). These data suggest and highlights the importance of continued influenza surveillance among equids for novel viruses.
Strong evidence is present that H3N8 EIV can cause infection occasionally in humans, in light of experimental studies and natural infection during outbreaks follow up when humans had got exposed to the infected animals. According to several scientists; suggesting continuous influenza surveillance as an important need to assess the genetic mutation and emergence of zoonotic EIV. The observational, experimental and historical data compels the premise that EIV infection occurs in man. In recent years, infections with EIV in humans have not presented signs of proper infection, considering this if the mutation rate in EIV H3N8 continuous it is very much possible that it could cause serious infections in man with clinical signs.
Though we have these observational and experimental studies evidence and data which supports the evidence of H3N8 EIVs being zoonotic for time being. But we must also be careful to conclude it as zoonotic influenza for equine H3N8 virus, because it might also be confounded by several factors including pre-existing antibodies against influenza strains other than H3N8 and non-specific assay inhibitors. Having all these finding it is also important to mention that H3N8 is very much capable to emerge as an epidemic zoonotic virus in future; due to high chances of mutation after being circulating in various mammalian species for last two decades.
Considering the economic impact of equine influenza on the equine industry both for sports and working purpose is huge. Estimating the global data analysis showed recently the impact have increased which highlights the importance of vaccination in the equine population routinely. We are thankful to one of our co-author (Muhammad Akib Warraich) for evaluating the economic impact of EIV and the importance of vaccination.
5. Conclusions
Equine influenza H3N8 virus has a wide epidemiology of spread in equines instead of control measures including vaccination and quarantine. In the last first decade this century several studies have reported the unpredictable nature of H3N8 EIVs highlighting the need of continuous surveillance for H3N8 in equines and humans in contact with them for novel and threatening mutations.
References
- ABDEL-MONEIM, A.S., ABDEL-GHANY, A.E. and SHANY, S.A., 2010. Isolation and characterization of highly pathogenic avian influenza virus subtype H5N1 from donkeys. Journal of Biomedical Science, vol. 17, no. 1, pp. 25. http://dx.doi.org/10.1186/1423-0127-17-25 PMid:20398268.
» http://dx.doi.org/10.1186/1423-0127-17-25 - ALDER, M., ed., 2008. Summary of the Australian equine influenza outbreak. The Veterinary Record, vol. 163, no. 13, pp. 378. http://dx.doi.org/10.1136/vr.163.13.378 PMid:19031639.
» http://dx.doi.org/10.1136/vr.163.13.378 - ALFORD, R.H., KASEL, J.A., LEHRICH, J.R. and KNIGHT, V., 1967. Human responses to experimental infection with influenza A/Equi 2 virus. American Journal of Epidemiology, vol. 86, no. 1, pp. 185-192. http://dx.doi.org/10.1093/oxfordjournals.aje.a120723 PMid:4951557.
» http://dx.doi.org/10.1093/oxfordjournals.aje.a120723 - ARTHUR, M., 2014. Institute for health metrics and evaluation. Nursing Standard, vol. 28, no. 42, pp. 32. http://dx.doi.org/10.7748/ns.28.42.32.s33 PMid:25005408.
» http://dx.doi.org/10.7748/ns.28.42.32.s33 - BACK, H., BERNDTSSON, L.T., GRÖNDAHL, G., STÅHL, K., PRINGLE, J. and ZOHARI, S., 2016. The first reported Florida clade 1 virus in the Nordic countries, isolated from a Swedish outbreak of equine influenza in 2011. Veterinary Microbiology, vol. 184, pp. 1-6. http://dx.doi.org/10.1016/j.vetmic.2015.12.010 PMid:26854337.
» http://dx.doi.org/10.1016/j.vetmic.2015.12.010 - BARQUERO, N., DALY, J.M. and NEWTON, J.R., 2007. Risk factors for influenza infection in vaccinated racehorses: lessons from an outbreak in Newmarket, UK in 2003. Vaccine, vol. 25, no. 43, pp. 7520-7529. http://dx.doi.org/10.1016/j.vaccine.2007.08.038 PMid:17889409.
» http://dx.doi.org/10.1016/j.vaccine.2007.08.038 - BEUTTEMMÜLLER, E.A., WOODWARD, A., RASH, A., FERRAZ, L.E.S., ALFIERI, A.F., ALFIERI, A.A. and ELTON, D., 2016. Characterisation of the epidemic strain of H3N8 equine influenza virus responsible for outbreaks in South America in 2012. Virology Journal, vol. 13, no. 1, pp. 45. http://dx.doi.org/10.1186/s12985-016-0503-9 PMid:26993620.
» http://dx.doi.org/10.1186/s12985-016-0503-9 - BRYANT, N.A., RASH, A.S., RUSSELL, C.A., ROSS, J., COOKE, A., BOWMAN, S., MACRAE, S., LEWIS, N.S., PAILLOT, R., ZANONI, R., MEIER, H., GRIFFITHS, L.A., DALY, J.M., TIWARI, A., CHAMBERS, T.M., NEWTON, J.R. and ELTON, D.M., 2009. Antigenic and genetic variations in European and North American equine influenza virus strains (H3N8) isolated from 2006 to 2007. Veterinary Microbiology, vol. 138, no. 1-2, pp. 41-52. http://dx.doi.org/10.1016/j.vetmic.2009.03.004 PMid:19346084.
» http://dx.doi.org/10.1016/j.vetmic.2009.03.004 - BURNELL, F.J., HOLMES, M.A., ROIKO, A.H., LOWE, J.B., HEIL, G.L., WHITE, S.K. and GRAY, G.C., 2014. Little evidence of human infection with equine influenza during the 2007 epizootic, queensland, australia. Journal of Clinical Virology, vol. 59, no. 2, pp. 100-103. http://dx.doi.org/10.1016/j.jcv.2013.11.011
» http://dx.doi.org/10.1016/j.jcv.2013.11.011 - CHEN, W., CALVO, P.A., MALIDE, D., GIBBS, J., SCHUBERT, U., BACIK, I., BASTA, S., O’NEILL, R., SCHICKLI, J., PALESE, P., HENKLEIN, P., BENNINK, J.R. and YEWDELL, J.W., 2001. A novel influenza A virus mitochondrial protein that induces cell death. Nature Medicine, vol. 7, no. 12, pp. 1306-1312. http://dx.doi.org/10.1038/nm1201-1306 PMid:11726970.
» http://dx.doi.org/10.1038/nm1201-1306 - CONNOR, R.J., KAWAOKA, Y., WEBSTER, R.G. and PAULSON, J.C., 1994. Receptor specificity in human, avian, and equine H2 and H3 influenza virus isolates. Virology, vol. 205, no. 1, pp. 17-23. http://dx.doi.org/10.1006/viro.1994.1615 PMid:7975212.
» http://dx.doi.org/10.1006/viro.1994.1615 - COUCEIRO, J.N.S., PAULSON, J.C. and BAUM, L.G., 1993. Influenza virus strains selectively recognize sialyloligosaccharides on human respiratory epithelium; the role of the host cell in selection of hemagglutinin receptor specificity. Virus Research, vol. 29, no. 2, pp. 155-165. http://dx.doi.org/10.1016/0168-1702(93)90056-S PMid:8212857.
» http://dx.doi.org/10.1016/0168-1702(93)90056-S - COUCH, R., DOUGLAS, R., KASEL, J., RIGGS, S. and KNIGHT, V., 1969. Production of the influenza syndrome in man with equine influenza virus. Nature, vol. 224, no. 5218, pp. 512-514. http://dx.doi.org/10.1038/224512a0
» http://dx.doi.org/10.1038/224512a0 - CRAWFORD, P.C., DUBOVI, E.J., CASTLEMAN, W.L., STEPHENSON, I., GIBBS, E., CHEN, L., SMITH, C., HILL, R.C., FERRO, P., POMPEY, J., BRIGHT, R.A., MEDINA, M.J., JOHNSON, C.M., OLSEN, C.W., COX, N.J., KLIMOV, A.I., KATZ, J.M. and DONIS, R.O., 2005. Transmission of equine influenza virus to dogs. Science, vol. 310, no. 5747, pp. 482-485. http://dx.doi.org/10.1126/science.1117950 PMid:16186182.
» http://dx.doi.org/10.1126/science.1117950 - CRISPE, E., FINLAISON, D., HURT, A. and KIRKLAND, P., 2011. Infection of dogs with equine influenza virus: evidence for transmission from horses during the Australian outbreak. Australian Veterinary Journal, vol. 89, suppl. 1, pp. 27-28. http://dx.doi.org/10.1111/j.1751-0813.2011.00734.x PMid:21711279.
» http://dx.doi.org/10.1111/j.1751-0813.2011.00734.x - CULLINANE, A., ELTON, D. and MUMFORD, J., 2010. Equine influenza–surveillance and control. Influenza and Other Respiratory Viruses, vol. 4, no. 6, pp. 339-344. http://dx.doi.org/10.1111/j.1750-2659.2010.00176.x PMid:20958927.
» http://dx.doi.org/10.1111/j.1750-2659.2010.00176.x - DALY, J.M., BLUNDEN, A.S., MACRAE, S., MILLER, J., BOWMAN, S.J., KOLODZIEJEK, J., NOWOTNY, N. and SMITH, K.C., 2008. Transmission of equine influenza virus to English foxhounds. Emerging Infectious Diseases, vol. 14, no. 3, pp. 461-464. http://dx.doi.org/10.3201/eid1403.070643 PMid:18325262.
» http://dx.doi.org/10.3201/eid1403.070643 - DALY, J.M., LAI, A., BINNS, M., CHAMBERS, T., BARRANDEGUY, M. and MUMFORD, J., 1996. Antigenic and genetic evolution of equine H3N8 influenza A viruses. The Journal of General Virology, vol. 77, no. Pt 4, pp. 661-671. http://dx.doi.org/10.1099/0022-1317-77-4-661 PMid:8627254.
» http://dx.doi.org/10.1099/0022-1317-77-4-661 - DALY, J.M., MACRAE, S., NEWTON, J.R., WATTRANG, E. and ELTON, D.M., 2011. Equine influenza: a review of an unpredictable virus. Veterinary Journal, vol. 189, no. 1, pp. 7-14. http://dx.doi.org/10.1016/j.tvjl.2010.06.026 PMid:20685140.
» http://dx.doi.org/10.1016/j.tvjl.2010.06.026 - DAMIANI, A.M., SCICLUNA, M., CIABATTI, I., CARDETI, G., SALA, M., VULCANO, G., CORDIOLI, P., MARTELLA, V., AMADDEO, D. and AUTORINO, G., 2008. Genetic characterization of equine influenza viruses isolated in Italy between 1999 and 2005. Virus Research, vol. 131, no. 1, pp. 100-105. http://dx.doi.org/10.1016/j.virusres.2007.08.001 PMid:17889395.
» http://dx.doi.org/10.1016/j.virusres.2007.08.001 - FOUCHIER, R.A., MUNSTER, V., WALLENSTEN, A., BESTEBROER, T.M., HERFST, S., SMITH, D., RIMMELZWAAN, G.F., OLSEN, B. and OSTERHAUS, A.D., 2005. Characterization of a novel influenza A virus hemagglutinin subtype (H16) obtained from black-headed gulls. Journal of Virology, vol. 79, no. 5, pp. 2814-2822. http://dx.doi.org/10.1128/JVI.79.5.2814-2822.2005 PMid:15709000.
» http://dx.doi.org/10.1128/JVI.79.5.2814-2822.2005 - FRANCIS JUNIOR, T., SALK, J.E., PEARSON, H.E. and BROWN, P.N., 1945. Protective effect of vaccination against induced influenza A. The Journal of Clinical Investigation, vol. 24, no. 4, pp. 536-546. http://dx.doi.org/10.1172/JCI101633 PMid:16695243.
» http://dx.doi.org/10.1172/JCI101633 - GARNER, M.G., COWLED, B., EAST, I., MOLONEY, B. and KUNG, N., 2011. Evaluating the effectiveness of early vaccination in the control and eradication of equine influenza: a modelling approach. Preventive Veterinary Medicine, vol. 99, no. 1, pp. 15-27. http://dx.doi.org/10.1016/j.prevetmed.2010.02.007 PMid:20236718.
» http://dx.doi.org/10.1016/j.prevetmed.2010.02.007 - GIBBS, E.P.J. and ANDERSON, T.C., 2010. Equine and canine influenza: a review of current events. Animal Health Research Reviews, vol. 11, no. 1, pp. 43-51. http://dx.doi.org/10.1017/S1466252310000046 PMid:20426896.
» http://dx.doi.org/10.1017/S1466252310000046 - GLASS, K., WOOD, J., MUMFORD, J., JESSET, D. and GRENFELL, B., 2002. Modelling equine influenza 1: a stochastic model of within-yard epidemics. Epidemiology and Infection, vol. 128, no. 3, pp. 491-502. http://dx.doi.org/10.1017/S0950268802006829 PMid:12113495.
» http://dx.doi.org/10.1017/S0950268802006829 - GORMAN, O.T., BEAN, W., KAWAOKA, Y., DONATELLI, I., GUO, Y. and WEBSTER, R., 1991. Evolution of influenza A virus nucleoprotein genes: implications for the origins of H1N1 human and classical swine viruses. Journal of Virology, vol. 65, no. 7, pp. 3704-3714. http://dx.doi.org/10.1128/jvi.65.7.3704-3714.1991 PMid:2041090.
» http://dx.doi.org/10.1128/jvi.65.7.3704-3714.1991 - GOTTSCHALK, A., BELYAVIN, G. and BIDDLE, F., 1972. Glycoproteins as influenza virus haemagglutinin inhibitors and as cellular virus receptors. In: A. Gottschalk, ed. Glycoproteins Amsterdam: Elsevier.
- GUO, Y., WANG, M., KAWAOKA, Y., GORMAN, O., ITO, T., SAITO, T. and WEBSTER, R.G., 1992. Characterization of a new avian-like influenza A virus from horses in China. Virology, vol. 188, no. 1, pp. 245-255. http://dx.doi.org/10.1016/0042-6822(92)90754-D PMid:1314452.
» http://dx.doi.org/10.1016/0042-6822(92)90754-D - GUTHRIE, A., 2006. Equine influenza in South Africa, 2003 outbreak. In: Proceedings of the Proceedings of the 9th International Congress of World Equine Veterinary Association, 22-26 January 2006, Marrakech, Morocco. Marrakech: World Equine Veterinary Association.
- GUTHRIE, A., STEVENS, K. and BOSMAN, P., 1999. The circumstances surrounding the outbreak and spread of equine influenza in South Africa (emergency management). In: Office international des épizooties, ed. Revue Scientifique et Technique de l'OIE. Paris: Office international des épizooties.
- HAMMOND, J., KEENEY, R. and RAIFFA, R., 2002. A practical guide to making better life decisions. New York: Broadway Books.
- HE, Q., 2012. Isolation and whole genome sequence analysis of equine H9N2 influenza virus in Guangxi Province, China Nanning, China: Guangxi University. Masters Dissertation.
- HORTON, R., 2013. Metrics for what? The Lancet, vol. 381, pp. S1-S2. http://dx.doi.org/10.1016/S0140-6736(13)61256-8
» http://dx.doi.org/10.1016/S0140-6736(13)61256-8 - JONG, N.K. and STONE, P., 2006. Kernel-Based Models for Reinforcement Learning. In: Proceedings of the ICML-06 Workshop on Kernel Methods in Reinforcement Learning, 25-29 January 2006, Pittsburgh, Pennsylvania, USA. New Jersey: John Wiley & Sons.
- KASEL, J.A. and COUCH, R., 1969. Experimental infections in man and horses with influenza A viruses. Bulletin of the World Health Organization, vol. 41, no. 3, pp. 447-452. PMid:5309454.
- KASEL, J.A., ALFORD, R.H., KNIGHT, V., WADDELL, G.H. and SIGEL, M.M., 1965. Experimental infection of human volunteers with equine influenza virus. Nature, vol. 206, no. 4979, pp. 41-43. http://dx.doi.org/10.1038/206041a0 PMid:14334358.
» http://dx.doi.org/10.1038/206041a0 - KAWAOKA, Y., BEAN, W.J. and WEBSTER, R.G., 1989. Evolution of the hemagglutinin of equine H3 influenza viruses. Virology, vol. 169, no. 2, pp. 283-292. http://dx.doi.org/10.1016/0042-6822(89)90153-0 PMid:2705299.
» http://dx.doi.org/10.1016/0042-6822(89)90153-0 - KHAN, A., MUSHTAQ, M.H., AHMAD, M.U.D., NAZIR, J., FAROOQI, S.H. and KHAN, A., 2017. Molecular Epidemiology of a novel re-assorted epidemic strain of equine influenza virus in Pakistan in 2015–16. Virus Research, vol. 240, pp. 56-63. http://dx.doi.org/10.1016/j.virusres.2017.07.022
» http://dx.doi.org/10.1016/j.virusres.2017.07.022 - KHAN, A., MUSHTAQ, M.H., AHMAD, M.U.D., NAZIR, J., FATIMA, Z., KHAN, A. and FAROOQI, S.H., 2018. Molecular epidemiology of the two internal genes of equine influenza H3N8 virus isolated in Pakistan 2015-16. Pakistan Veterinary Journal, vol. 38, no. 1, pp. 96-100.
- KHURELBAATAR, N., KRUEGER, W.S., HEIL, G.L., DARMAA, B., ULZIIMAA, D., TSERENNOROV, D., BATERDENE, A., ANDERSON, B.D. and GRAY, G.C., 2013. Sparse evidence for equine or avian influenza virus infections among Mongolian adults with animal exposures. Influenza and Other Respiratory Viruses, vol. 7, no. 6, pp. 1246-1250. http://dx.doi.org/10.1111/irv.12148 PMid:23941547.
» http://dx.doi.org/10.1111/irv.12148 - KHURELBAATAR, N., KRUEGER, W.S., HEIL, G.L., DARMAA, B., ULZIIMAA, D., TSERENNOROV, D., BATERDENE, A., ANDERSON, B.D. and GRAY, G.C., 2014. Little evidence of avian or equine influenza virus infection among a cohort of mongolian adults with animal exposures, 2010–2011. PLoS One, vol. 9, no. 1, pp. e85616. http://dx.doi.org/10.1371/journal.pone.0085616
» http://dx.doi.org/10.1371/journal.pone.0085616 - KING, E. and MACDONALD, D., 2004. Report of the Board of Inquiry appointed by the Board of the National Horseracing Authority to conduct enquiry into the causes of the equine influenza which started in the Western cape in early December 2003 and spread to the Eastern Cape and Gauteng. Australian Equine Veterinarian, vol. 23, pp. 139-142.
- KIRKLAND, P.D., FINLAISON, D.S., CRISPE, E. and HURT, A.C., 2010. Influenza virus transmission from horses to dogs, Australia. Emerging Infectious Diseases, vol. 16, no. 4, pp. 699-702. http://dx.doi.org/10.3201/eid1604.091489 PMid:20350392.
» http://dx.doi.org/10.3201/eid1604.091489 - KIRKWOOD, C., 1998. Strategic decision making multiobjective decision analysis with spreadsheets. The Journal of the Operational Research Society, vol. 49, no. 1, pp. 96-97. http://dx.doi.org/10.1057/palgrave.jors.2600003
» http://dx.doi.org/10.1057/palgrave.jors.2600003 - KRUEGER, W.S., HEIL, G.L., YOON, K.J. and GRAY, G.C., 2014. No evidence for zoonotic transmission of H 3 N 8 canine influenza virus among US adults occupationally exposed to dogs. Influenza and Other Respiratory Viruses, vol. 8, no. 1, pp. 99-106. http://dx.doi.org/10.1111/irv.12208 PMid:24237615.
» http://dx.doi.org/10.1111/irv.12208 - LANDOLT, G.A., 2014. Equine influenza virus. Veterinary Clinics of North America: Equine Practice, vol. 30, no. 3, pp. 507-522. PMid:25282321.
- LARSON, K.R.L., HEIL, G.L., CHAMBERS, T.M., CAPUANO, A., WHITE, S.K. and GRAY, G.C., 2015. Serological evidence of equine influenza infections among persons with horse exposure, Iowa. Journal of Clinical Virology, vol. 67, pp. 78-83. http://dx.doi.org/10.1016/j.jcv.2015.04.009 PMid:25959164.
» http://dx.doi.org/10.1016/j.jcv.2015.04.009 - LEGRAND, L.J, PITEL, P.H., MARCILLAUD‐PITEL, C., CULLINANE, A., COUROUCE, A., FORTIER, G., FREYMUTH, F. and PRONOST, S., 2013. Surveillance of equine influenza viruses through the RESPE network in France from November 2005 to October 2010. Equine Veterinary Journal, vol. 45, no. 6, pp. 776-783. http://dx.doi.org/10.1111/evj.12100 PMid:23662725.
» http://dx.doi.org/10.1111/evj.12100 - LOWEN, A. and PALESE, P., 2009. Transmission of influenza virus in temperate zones is predominantly by aerosol, in the tropics by contact: a hypothesis. PLoS Currents, vol. 1, p. RRN1002. http://dx.doi.org/10.1371/currents.RRN1002 PMid:20025197.
» http://dx.doi.org/10.1371/currents.RRN1002 - MORENS, D.M. and TAUBENBERGER, J.K., 2010. An avian outbreak associated with panzootic equine influenza in 1872: an early example of highly pathogenic avian influenza? Influenza Other Respi. Viruses, vol. 4, pp. 373-377.
- MORENS, D.M. and TAUBENBERGER, J.K., 2010a. An avian outbreak associated with panzootic equine influenza in 1872: an early example of highly pathogenic avian influenza? Influenza and Other Respiratory Viruses, vol. 4, no. 6, pp. 373-377. http://dx.doi.org/10.1111/j.1750-2659.2010.00181.x PMid:20958931.
» http://dx.doi.org/10.1111/j.1750-2659.2010.00181.x - NEMOTO, M., YAMANAKA, T., BANNAI, H., TSUJIMURA, K., KONDO, T. and MATSUMURA, T., 2012. Development of a reverse transcription loop-mediated isothermal amplification assay for H7N7 equine influenza virus. The Journal of Veterinary Medical Science, vol. 74, no. 7, pp. 929-931. http://dx.doi.org/10.1292/jvms.11-0541 PMid:22362254.
» http://dx.doi.org/10.1292/jvms.11-0541 - NEWTON, J.R, DALY, J., SPENCER, L. and MUMFORD, J., 2006. Description of the outbreak of equine influenza (H3N8) in the United Kingdom in 2003, during which recently vaccinated horses in Newmarket developed respiratory disease. The Veterinary Record, vol. 158, no. 6, pp. 185-192. http://dx.doi.org/10.1136/vr.158.6.185 PMid:16474051.
» http://dx.doi.org/10.1136/vr.158.6.185 - NEWTON, R., COOKE, A., ELTON, D., BRYANT, N., RASH, A., BOWMAN, S., BLUNDEN, T., MILLER, J., HAMMOND, T.-A., CAMM, I. and DAY, M., 2007. Canine influenza virus: cross-species transmission from horses. The Veterinary Record, vol. 161, no. 4, pp. 142-143. http://dx.doi.org/10.1136/vr.161.4.142-a PMid:17660470.
» http://dx.doi.org/10.1136/vr.161.4.142-a - PARRISH, C.R., MURCIA, P.R. and HOLMES, E.C., 2015. Influenzavirusreservoirsandintermediatehosts: Dogs, horses, and new possibilities for influenza virus exposure of humans. Journal of Virology, vol. 89, no. 6, pp. 2990-2994. http://dx.doi.org/10.1128/JVI.03146-14
» http://dx.doi.org/10.1128/JVI.03146-14 - RAIFFA, H., HAMMOND JOHN, S. and KEENEY RALPH, L., 1999. Smart choices: a practical guide to making better life decisions. Boston: Harvard Business School Press.
- ROZEK, W., PURZYCKA, M., POLAK, M.P., GRADZKI, Z. and ZMUDZINSKI, J.F., 2009. Genetic typing of equine influenza virus isolated in Poland in 2005 and 2006. Virus Research, vol. 145, no. 1, pp. 121-126. http://dx.doi.org/10.1016/j.virusres.2009.06.017 PMid:19580828.
» http://dx.doi.org/10.1016/j.virusres.2009.06.017 - SHI, W., LEI, F., ZHU, C., SIEVERS, F. and HIGGINS, D.G., 2010. A complete analysis of HA and NA genes of influenza A viruses. PLoS One, vol. 5, no. 12, pp. e14454. http://dx.doi.org/10.1371/journal.pone.0014454 PMid:21209922.
» http://dx.doi.org/10.1371/journal.pone.0014454 - STUDDERT, M.J., 1996. Virus infections of equines Amsterdam: Elsevier.
- SU, S., WANG, L., FU, X., HE, S., HONG, M., ZHOU, P., LAI, A., GRAY, G. and LI, S., 2014. Equine influenza A (H3N8) virus infection in cats. Emerging Infectious Diseases, vol. 20, no. 12, pp. 2096-2099. http://dx.doi.org/10.3201/eid2012.140867 PMid:25417790.
» http://dx.doi.org/10.3201/eid2012.140867 - SUZUKI, Y., ITO, T., SUZUKI, T., HOLLAND JUNIOR, R.E., CHAMBERS, T.M., KISO, M., ISHIDA, H. and KAWAOKA, Y., 2000. Sialic acid species as a determinant of the host range of influenza A viruses. Journal of Virology, vol. 74, no. 24, pp. 11825-11831. http://dx.doi.org/10.1128/JVI.74.24.11825-11831.2000 PMid:11090182.
» http://dx.doi.org/10.1128/JVI.74.24.11825-11831.2000 - TU, J., ZHOU, H., JIANG, T., LI, C., ZHANG, A., GUO, X., ZOU, W., CHEN, H. and JIN, M., 2009. Isolation and molecular characterization of equine H3N8 influenza viruses from pigs in China. Archives of Virology, vol. 154, no. 5, pp. 887-890. http://dx.doi.org/10.1007/s00705-009-0381-1 PMid:19396578.
» http://dx.doi.org/10.1007/s00705-009-0381-1 - VAN MAANEN, C. and CULLINANE, A., 2002. Equine influenza virus infections: an update. The Veterinary Quarterly, vol. 24, no. 2, pp. 79-94. http://dx.doi.org/10.1080/01652176.2002.9695127 PMid:12095083.
» http://dx.doi.org/10.1080/01652176.2002.9695127 - VIRMANI, N., BERA, B., SINGH, B., SHANMUGASUNDARAM, K., GULATI, B., BARUA, S., VAID, R., GUPTA, A. and SINGH, R., 2010. Equine influenza outbreak in India (2008–09): virus isolation, sero-epidemiology and phylogenetic analysis of HA gene. Veterinary Microbiology, vol. 143, no. 2-4, pp. 224-237. http://dx.doi.org/10.1016/j.vetmic.2009.12.007 PMid:20053509.
» http://dx.doi.org/10.1016/j.vetmic.2009.12.007 - WISE, H.M., FOEGLEIN, A., SUN, J., DALTON, R.M., PATEL, S., HOWARD, W., ANDERSON, E.C., BARCLAY, W.S. and DIGARD, P., 2009. A complicated message: identification of a novel PB1-related protein translated from influenza A virus segment 2 mRNA. Journal of Virology, vol. 83, no. 16, pp. 8021-8031. http://dx.doi.org/10.1128/JVI.00826-09 PMid:19494001.
» http://dx.doi.org/10.1128/JVI.00826-09 - YADAV, M., UPPAL, P. and MUMFORD, J., 1993. Physico-chemical and biological characterization of A/Equl-2 virus isolated from 1987 equine influenza epidemic in India. International Journal of Animal Sciences, vol. 8, pp. 93-98.
- YAMANAKA, T., NEMOTO, M., TSUJIMURA, K., KONDO, T. and MATSUMURA, T., 2009. Interspecies transmission of equine influenza virus (H3N8) to dogs by close contact with experimentally infected horses. Veterinary Microbiology, vol. 139, no. 3-4, pp. 351-355. http://dx.doi.org/10.1016/j.vetmic.2009.06.015 PMid:19596528.
» http://dx.doi.org/10.1016/j.vetmic.2009.06.015 - YONDON, M., HEIL, G.L., BURKS, J.P., ZAYAT, B., WALTZEK, T.B., JAMIYAN, B.O., MCKENZIE, P.P., KRUEGER, W.S., FRIARY, J.A. and GRAY, G.C., 2013. Isolation and characterization of H 3 N 8 equine influenza A virus associated with the 2011 epizootic in M ongolia. Influenza and Other Respiratory Viruses, vol. 7, no. 5, pp. 659-665. http://dx.doi.org/10.1111/irv.12069 PMid:23289427.
» http://dx.doi.org/10.1111/irv.12069 - YONDON, M., ZAYAT, B., NELSON, M.I., HEIL, G.L., ANDERSON, B.D., LIN, X., HALPIN, R.A., MCKENZIE, P.P., WHITE, S.K., WENTWORTH, D.E. and GRAY, G.C., 2014. Equine influenza A (H3N8) virus isolated from Bactrian camel, Mongolia. Emerging Infectious Diseases, vol. 20, no. 12, pp. 2144-2147. http://dx.doi.org/10.3201/eid2012.140435 PMid:25418532.
» http://dx.doi.org/10.3201/eid2012.140435
Publication Dates
-
Publication in this collection
27 Aug 2021 -
Date of issue
2023
History
-
Received
14 Dec 2020 -
Accepted
15 Feb 2021